Original Papers
2026
- Harnessing Random Polymers as Chemoselective Catalysts With Structural Adaptability: Epoxidation of Olefinic Quaternary Ammonium Salts via Charge Recognition
Fujii, Y.; Takasu, K.; Kuroda, Y.
Chem. Eur. J. 2026, xxx, xxx-xxx.
DOI: 10.1002/chem.71082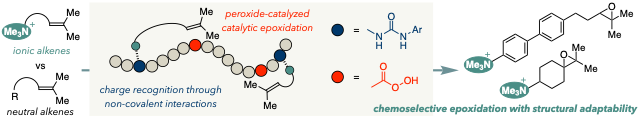
- One-Pot Multi-Substrate Screening of Ligation Reactions Using PNA Tags
Kohyama, A.; Barluenga, S.; Winssinger, N.
Chem. Sci. 2026, xxx, xxx-xxx.
DOI: 10.1039/d5sc08732e
- Toward N-Linked Glycoproteins: Three-Step Synthesis of Glycan-Asparagine Conjugates from Unprotected Sugars
Akeno, J.; Takasu, K.; Oisaki, K.; Kumada-Nozawa, K.; Sada, H.; Nanjo, T.; Fukaya, N.; Ueda, Y.
Org. Lett. 2026, 28, 3441-3447.
DOI: 10.1021/acs.orglett.6c00314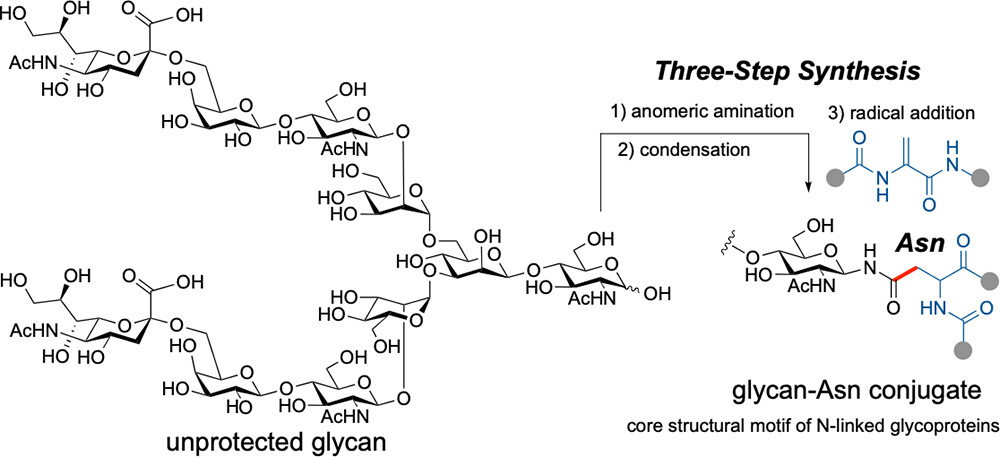
- Self-Induced Charge Transfer Activation Enables Metal-Free C–H Coupling of Polycyclic Aromatic Hydrocarbons under Photo Irradiation
Kurokawa, K.; Kohyama, A.; Kuroda, Y.; Tao-Kakuyama, K.; Takikawa, H.; Takasu, K.
Chem. Sci. 2026, 17, 4942-4947.
DOI: 10.1039/d5sc09144f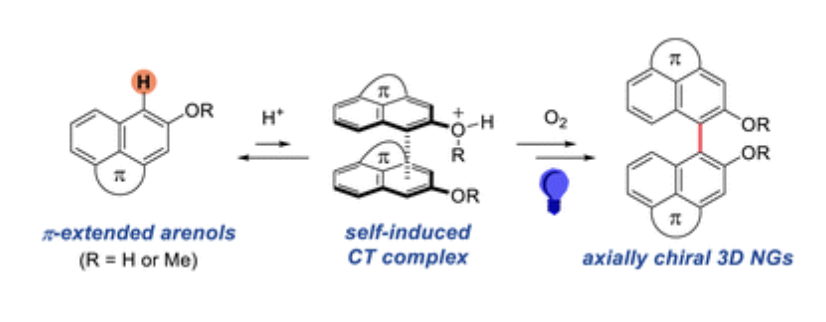
2025
- Synthesis and Properties of Medium-Sized cis,trans-Cycloocta-1,3-diene–Pt(II) Complexes
Kinouchi, H.; Ito, T.; Kohyama, A.; Kuroda, Y.; Takikawa, H.; Takasu, K.
Org. Lett. 2025, 27, 13922-13927.
DOI: 10.1021/acs.orglett.5c04468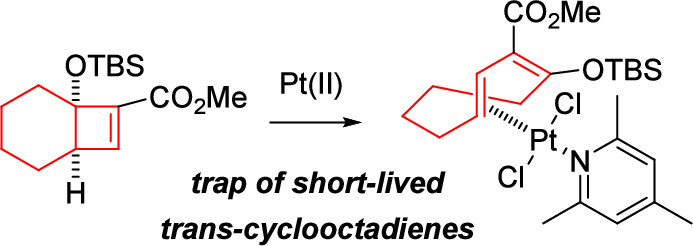
- Net δ-C–H Alkylation of Allyl Alcohols by Ni-Catalyzed Allylic Substitution of Azo-Ene Adducts
Kawajiri, M.; Kuroda, Y.; Takasu, K.
J. Org. Chem. 2025, 90, 12826-12831.
DOI: 10.1021/acs.joc.5c01497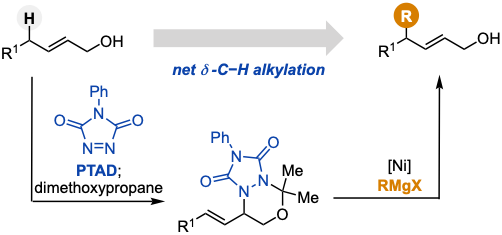
- A Potassium Base-Promoted Intramolecular Enolate–Olefin Metathesis
Sugimoto, K.; Nagao, T.; Kurokawa, K.; Tawatari, T.; Miyakawa, Y.; Fujimura, S.; Yamada, K.; Yamaoka, Y.; Kohyama, A.; Kuroda, Y.; Takikawa, H.; Takasu, K.
Chem. Eur. J. 2025, 31, e00737.
DOI: 10.1002/chem.202500737
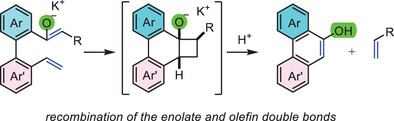
- Unveiling the Inverse-Handed Self-Discrimination of Helicenes with Twisted π-Surfaces by Liquid Chromatography
Kanao, E.; Murata, Y.; Miwa, S.; Takikawa, H.; Takasu, K.; Ishihama, Y.; Kubo, T.
Anal. Chem. 2025, 97, 12690-12698.
DOI: 10.1021/acs.analchem.5c01385
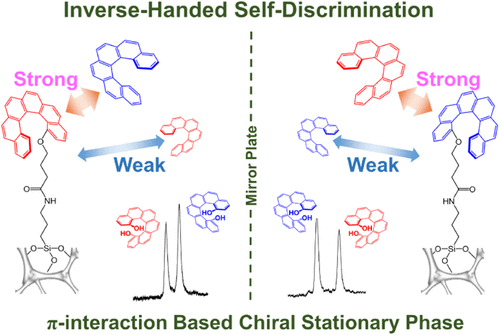
- Arylative Double Bond Transposition of Allylic Alcohols via Silicon-Tethered Intramolecular Benzyne–Ene Reactions
Kato, R.; Sakaue, T.; Tawatari, T.; Takasu, K.; Takikawa, H.
Chem. Commun. 2025, 61, 8347-8350.
DOI: 10.1039/D5CC01918D
Selected as Chemical Communications HOT articles 2025
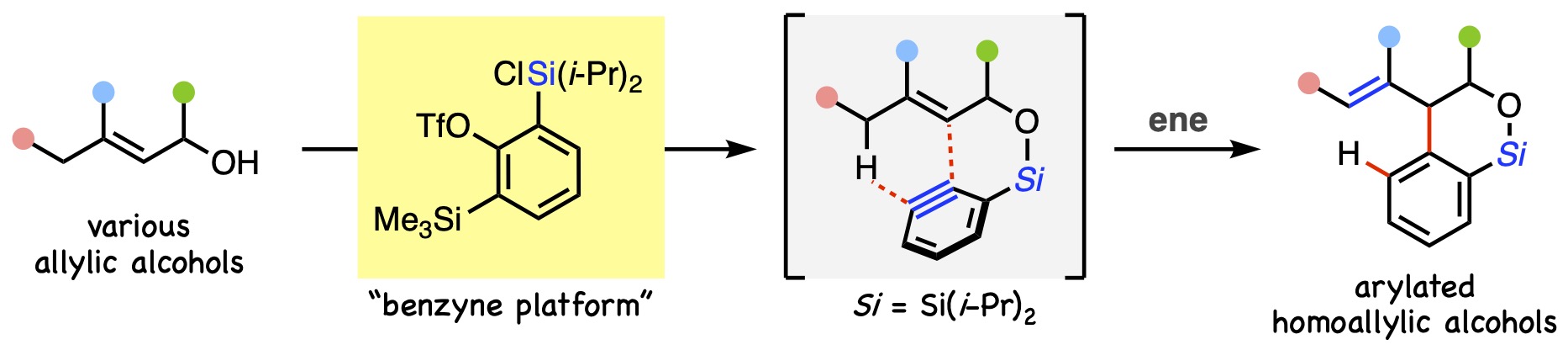
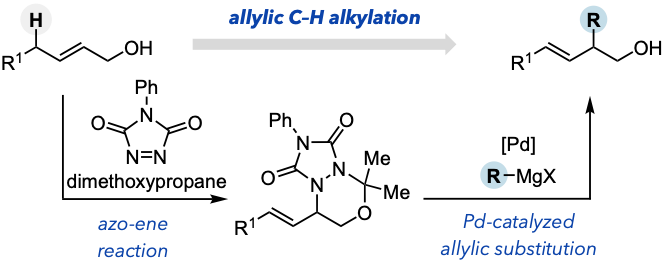
- Pd-Catalyzed Allylic Substitution of Azo-Ene Adducts Enables Net Allylic C–H Alkylation of Allylic Alcohols
Kuroda, Y.; Chiba, T.; Kawajiri, M.; Takasu, K.
Org. Lett. 2025, 27, 1517-1523.
DOI: 10.1021/acs.orglett.5c00049
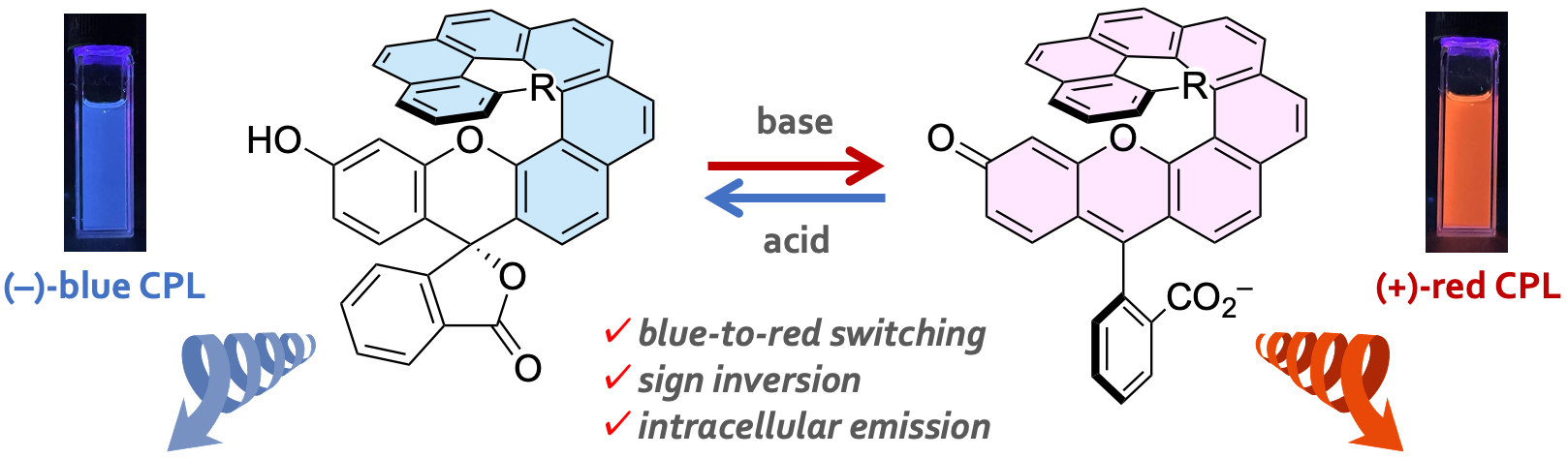
- Helicene–Fluorescein Hybrids: A Reversible Base-triggered (Chir)optical Switch with Sign Inversion of Circularly Polarized Luminescence
Miwa, S.; Mizutani, D.; Kawano, K.; Matsuzaki, K.; Nagata, Y.; Tsubaki, K.; Takasu, K.; Takikawa, H.
Chem. Eur. J. 2025, 31, e202500335.
DOI: 10.1002/chem.202500335
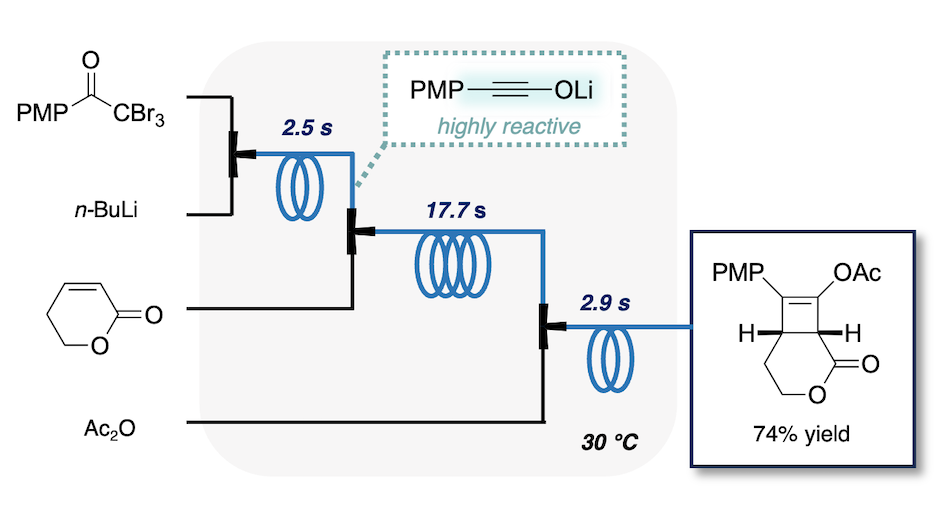
- Continuous Flow Synthesis of Cyclobutenes via Lithium Ynolates
Kohyama, A.; Namioka, M.; Naka, H.; Ashikari, Y.; Nagaki, A.; Takikawa, H.; Yamaoka, Y.; Takasu, K.
Green Chem. 2025, 27, 2760-2765.
DOI: 10.1039/D4GC05102E
2024
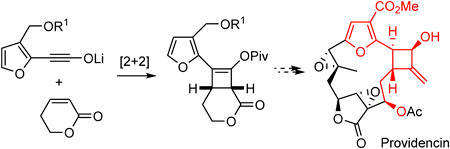
- Synthetic Study towards Providencin: Stereocontrolled Synthesis of the Furan-Substituted Cyclobutanol Segment
Yamaoka, Y.; Nishina, R.; Fujita, K.; Takasu, K.
Chem. Pharm. Bull. 2024, 72, 966-969.
DOI: 10.1248/cpb.c24-00629
Selected as a featured article
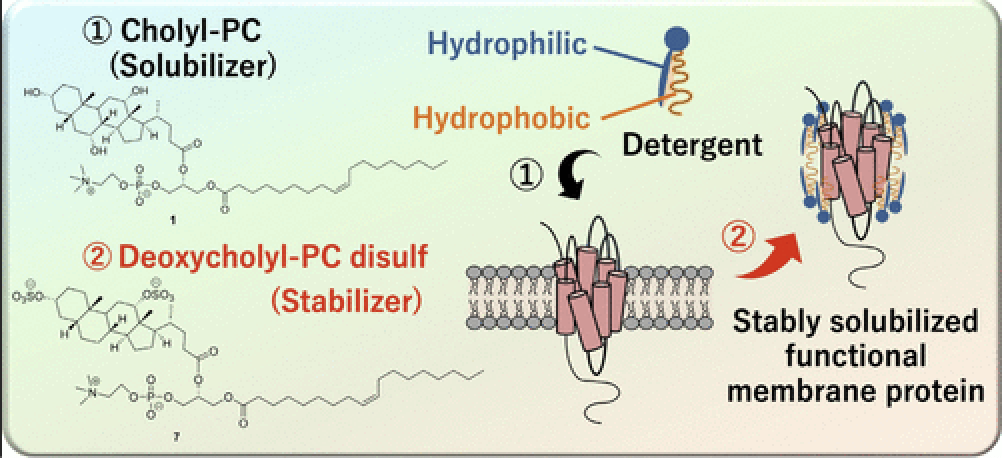
- Stable and Minimum Size Solubilization of Membrane Proteins with Cocktails of Phospholipid Analogues
Takagi, M.; Nagatani, A.; Kawano, K.; Hata, A.; Yokoyama, A.; Hayashida, K.; Hoshi, H.; Sakurai, M.; Oyama, T.; Kuroda, Y.; Yamaoka, Y.; Fujiwara, T.; Miyanoiri, Y.; Hoshino, M.; Yano, Y.; Takasu, K.; Matsuzaki, K
ACS Appl. Mater. Interfaces 2024, 16, 63358-63367.
DOI: 10.1021/acsami.4c15697
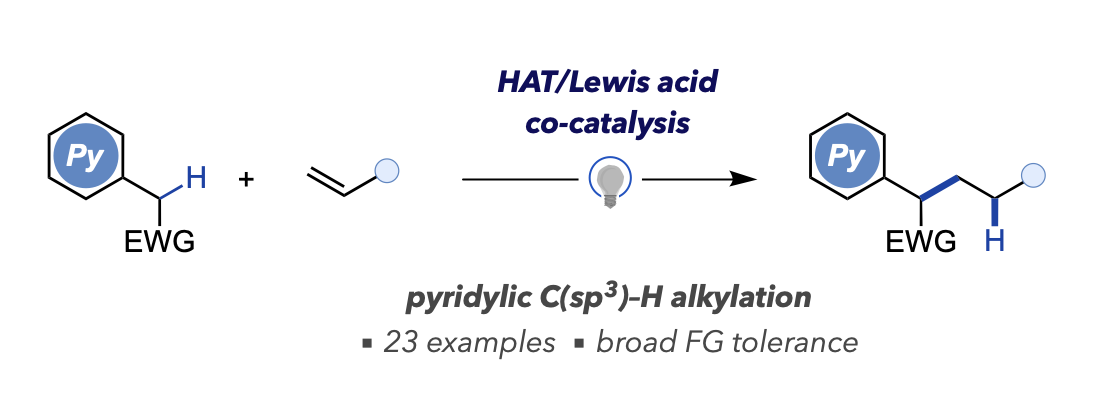
- Photo-Induced Pyridylic C(sp3)–H Alkylation with Unactivated Alkenes Enabled by Hydrogen Atom Transfer/Lewis Acid Cocatalysis
Kuroda, Y.; Saito, H.; Tawatari, T.; Takasu, K.
ACS Catal. 2024, 14, 15036-15042.
DOI: 10.1021/acscatal.4c05026
- Chromic Properties of Dibenzo[j,l]fluoranthenes Exhibiting Different Resonance Contributions
Kurokawa, K.; Ogawa, N.; Kuroda, Y.; Yamaoka, Y.; Takikawa, H.; Tsubaki, K.; Takasu, K.
Org. Biomol. Chem. 2024, 22, 5306-5313.
DOI: 10.1039/D4OB00750F

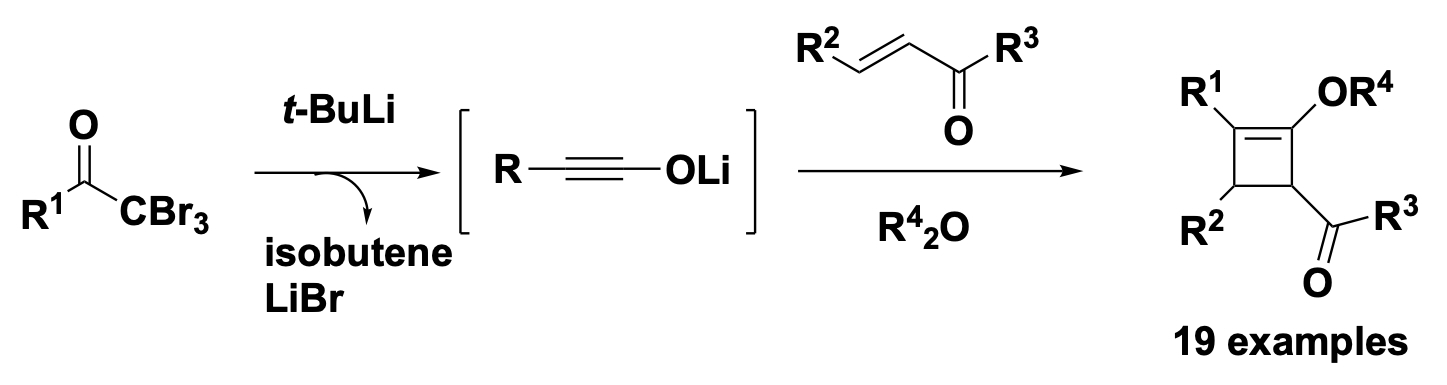
- Entry into Lithium Ynolates from α,α,α-Tribromomethyl Ketones: Synthesis of Cyclobutenes via the [2 + 2] Cycloaddition with α,β-Unsaturated Carbonyls
Yamaoka, Y.; Imahori, H.; Namioka, M.; Nishina, R.; Kobori, Y.; Ueda, M.; Shindo, M.; Takasu, K.
Org. Lett. 2024, 26, 1986-1901.
DOI: 10.1021/acs.orglett.4c00202
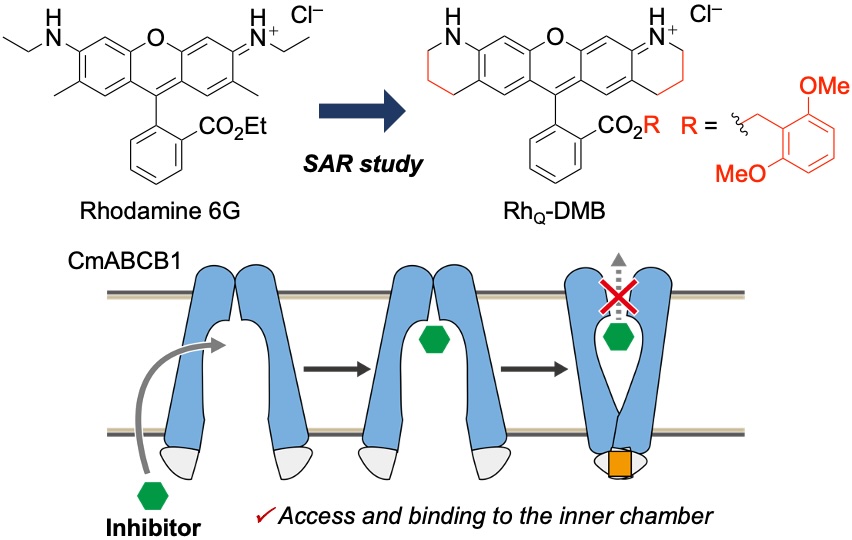
- Structure–ATPase Activity Relationship of Rhodamine Derivatives as Potent Inhibitors of P-Glycoprotein CmABCB1
Miwa, S.; Takikawa, H.; Takeuchi, R.; Mizunuma, R.: Matsuoka, K.; Ogawa, H.; Kato, H.; Takasu, K.
ACS Med. Chem. Lett. 2024, 15, 287-293.
DOI: 10.1021/acsmedchemlett.3c00526
- Synthesis of mesoionic triazolones via a formal [3+2] cycloaddition between 4-phenyl-1,2,4-triazoline-3,5-dione and alkynes
Kuroda, Y.; Krell, M.; Kurokawa, K.; Takasu, K.
Chem. Commun. 2024, 60, 1719-1722.
DOI: 10.1039/D3CC05088B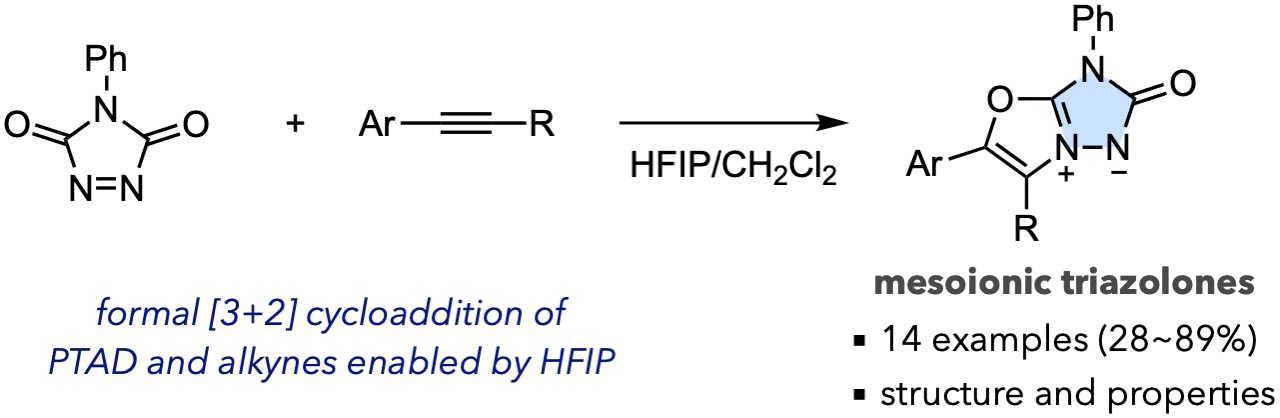
2023
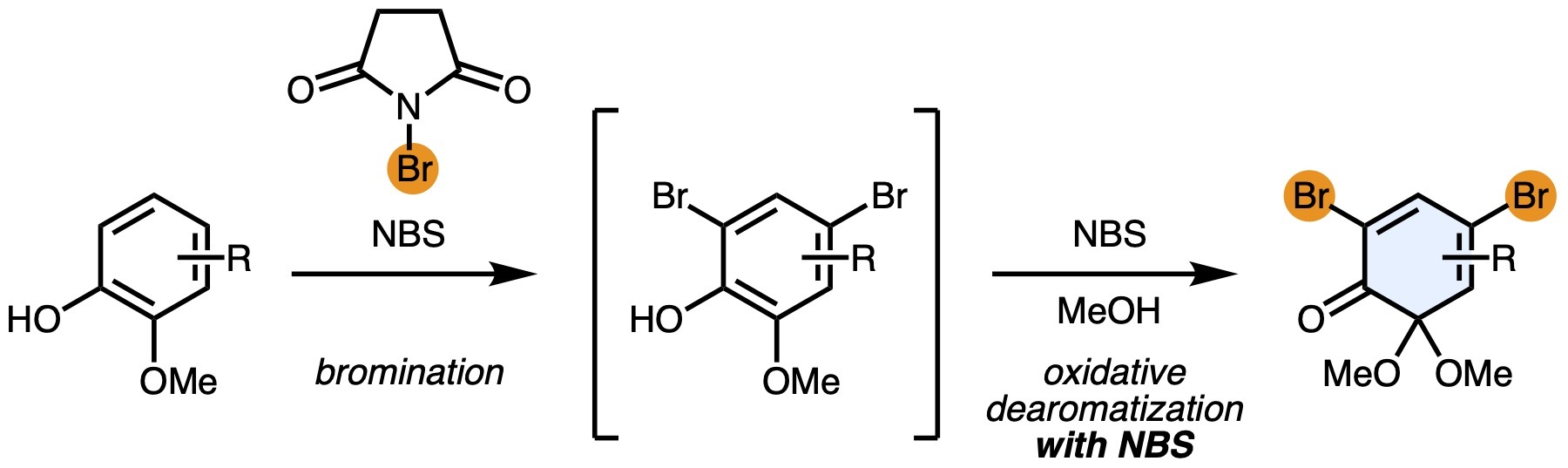
- Sequential Bromination/Dearomatization Reactions of 2-Methoxyphenols en route to Bromo-Substituted ortho-Quinonemonoacetals
Hayashi, S.; Takasu, K.; Takikawa, H.
Synlett 2023, 35, 474-478.
DOI: 10.1055/s-0041-1738450
- Rotaxane Synthesis by an End-Capping Strategy via Swelling Axle-Phenols
Fujimura, K.; Ueda, Y.; Yamaoka, Y.; Takasu, K.; Kawabata, T.
Angew. Chem. Int. Ed. 2023, 63, e202303078.
DOI: 10.1002/anie.202303078
- An Iridium/Aluminum Cooperative Strategy for the β-C(sp3)–H Borylation of Saturated Cyclic Amines
Kuroda, Y.; Park, K.; Shimazaki, Y.; Zhong, R.-L.; Sakaki, S.; Nakao, Y.
Angew. Chem. Int. Ed. 2023, 62, e202300704.
DOI: 10.1002/anie.202300704
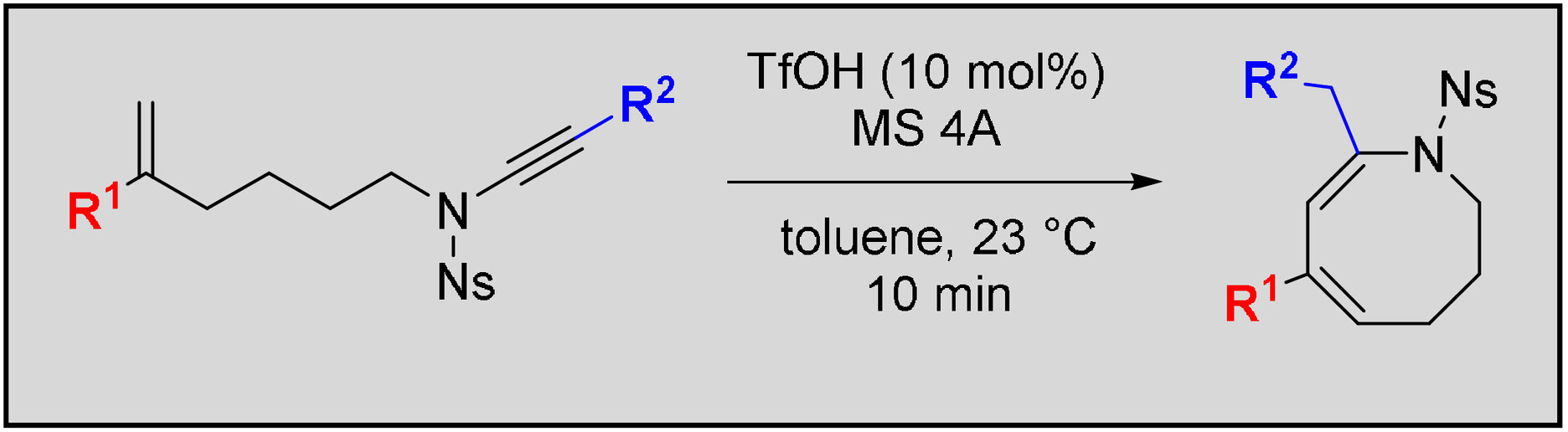
- Efficient Synthesis of Medium-sized Nitrogen Heterocycles by Brønsted Acid-Catalyzed Cyclization of Ene-ynamides
Yamaoka, Y.; Takeuchi, N.; Yamada, K.; Takasu, K.
Asian J. Org. Chem. 2023, 12, e202300145.
DOI: 10.1002/ajoc.202300145
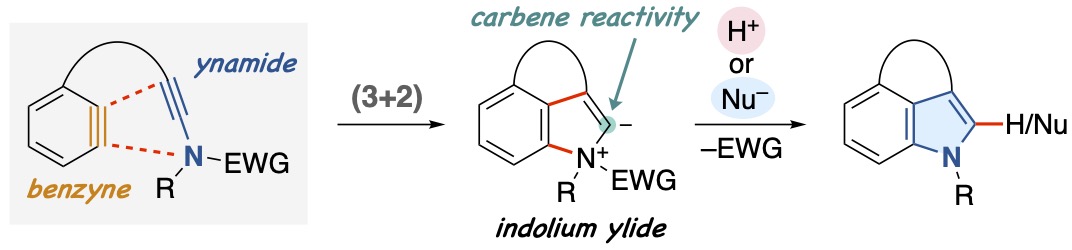
- Intramolecular Ynamide–Benzyne (3+2) Cycloadditions
Tawatari, T.; Kato, R.; Kudo, R.; Takasu, K.; Takikawa, H.
Angew. Chem. Int. Ed. 2023, 62, e202300907.
DOI: 10.1002/anie.202300907
京都大学プレスリリース
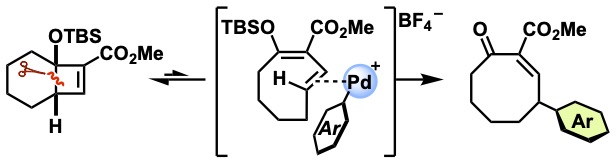
- Synthesis of γ-Aryl Medium-sized Cyclic Enones by a domino 4π-Electrocyclic Reaction—Heck–Matsuda Arylation Sequence at Ambient Temperature
Ito, T.; Takeuchi, N.; Yamaoka, Y.; Takikawa, H.; Takasu, K.
Synthesis 2023, 34, 1275-1279.
DOI: 10.1055/a-2024-4675
selected as the cover picture
2022
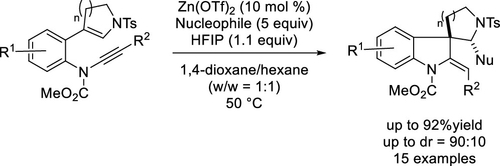
- Lewis Acid-Catalyzed Diastereoselective Domino Reaction of Ene-Ynamide with Trimethylsilyl Cyanide to Construct Spiroindolines
Yamaoka, Y.; Yamasaki, D.; Kajiwara, D.; Shinosaki, M.; Yamada, K.; Takasu, K.
Org. Lett. 2022, 24, 4389-4393.
DOI: 10.1021/acs.orglett.2c01607
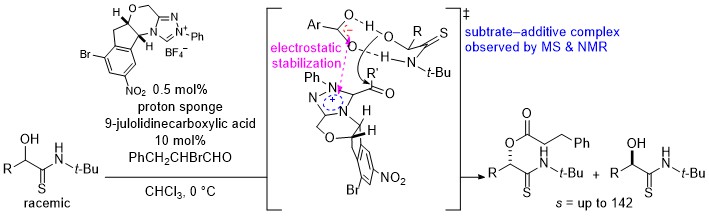
- Enhanced Molecular Recognition through Substrate–Additive Complex Formation in N-Heterocyclic-Carbene-Catalyzed Kinetic Resolution of α-Hydroxythioamides
Wang, Y.; Yamauchi, A.; Hashimoto, K.; Fujiwara, T.; Inokuma, T.; Mitani, Y.; Ute, K.; Kuwano, S.; Yamaoka, Y.; Takasu, K.; Yamada, K.
ACS Catal. 2022, 12, 6100-6107.
DOI: 10.1021/acscatal.2c01579
- Intramolecular Propargylic Ene Reaction of Benzyne en Route to Highly Functionalized Allenes and Allenamides
Tawatari, T.; Kato, R.; Takasu, K.; Takikawa, H.
Synthesis 2022, 54, 4979-4988.
DOI: 10.1055/a-1826-2545 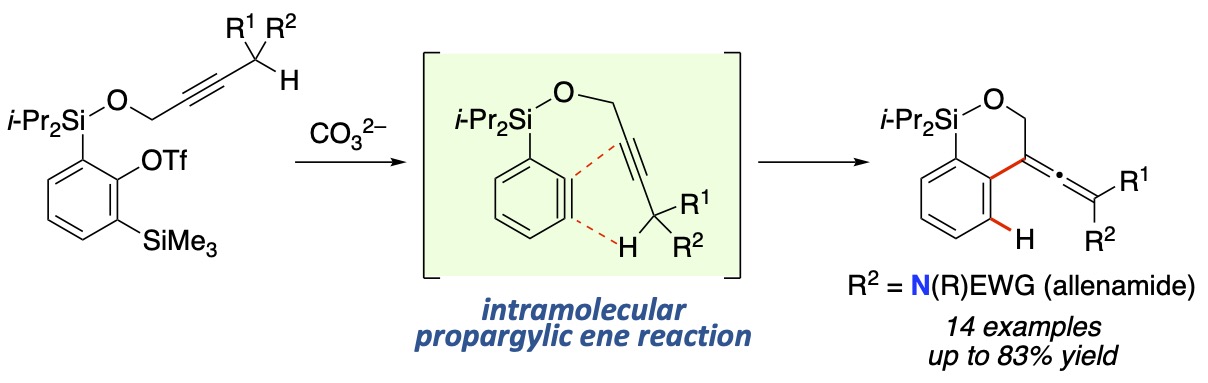
- AI-Driven Synthetic Route Design Incorporated with Retrosynthesis Knowledge
Ishida, S.; Terayama, K.; Kojima, R.; Takasu, K.; Okuno, Y.
J. Chem. Inf. Model. 2022, 62, 1357-1367.
DOI: 10.1021/acs.jcim.1c01074
- Total Synthesis of Cryptopleurine and Its Analogues
Yamaoka, Y.; Yamakawa, T.; Tateishi, K.; Takasu, K.
Synthesis 2022, 54, 2415-2422.
DOI: 10.1055/a-1730-8628 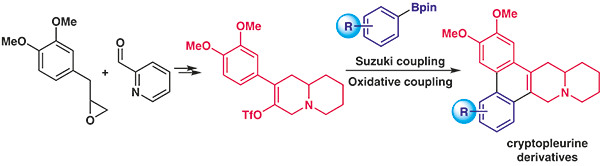
- Catalytic Substrate-Selective Silylation of Primary Alcohols via Remote Functional-Group Discrimination
Hashimoto, H.; Ueda, Y.; Takasu, K.; Kawabata, T.
Angew. Chem. Int. Ed. 2022, 61, e202114118.
DOI: 10.1002/anie.202114118
2021
- 2-(Chlorodiisopropylsilyl)-6-(trimethylsilyl)phenyl triflate: a modified platform for intramolecular benzyne cycloadditions
Tawatari, T.; Takasu, K.; Takikawa, H.
Chem. Commun. 2021, 57, 11863-11866.
DOI: 10.1039/D1CC05264K 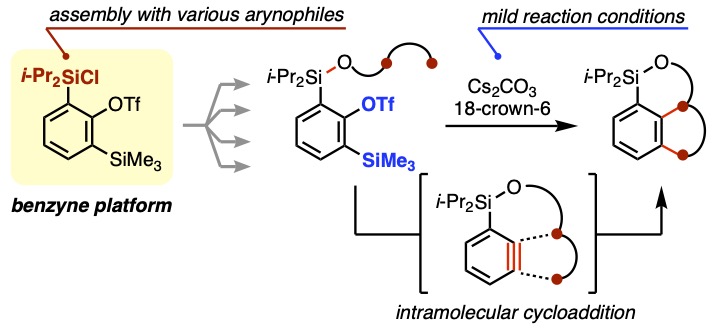
- Oxidative β-Cleavage of Fused Cyclobutanols Leading to Hydrofuran-Fused Polycyclic Aromatic Compounds
Kinouchi, H.; Sugimoto, K.; Yamaoka, Y.; Takikawa, H.; Takasu, K.
J. Org. Chem. 2021, 86, 12615-12622.
DOI: 10.1021/acs.joc.1c01108 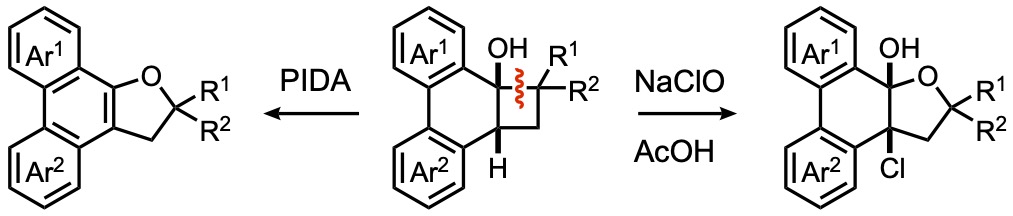
- The Rationale for Stereoinduction in Conjugate Addition to Alkylidenemalonates Bearing a Menthol-derived Chiral Auxiliary
Yamada, K.; Fujiwara, S.; Inokuma, T.; Sugano, M.; Yamaoka, Y.; Takasu, K.
Tetrahedron 2021, 91, 132220.
DOI: 10.1016/j.tet.2021.132220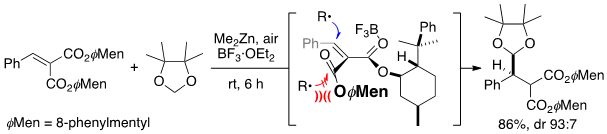
- Mechanistic Support for Intramolecular Migrative Cyclization of Propargyl Sulfones Provided by Catalytic Asymmetric Induction with a Chiral Counter Cation Strategy
Yamasaki, K.; Yamauchi, A.; Inokuma, T.; Miyakawa, Y.; Wang, Y.; Oriez, R.; Yamaoka, Y.; Takasu, K.; Tanaka, M.; Kashiwada, Y.; Yamada, K.
Asian J. Org. Chem. 2021, 10, 1828-1834.
DOI: 10.1002/ajoc.202100274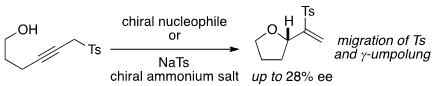
- Synthesis of Lactone-fused Cyclopropanes by Ring Contractive a-Ketol Rearrangement of Ketal-fused Cyclobutanones
Takasu, K.; Shigenaga, K.; Shimoda, K.; Takikawa, H.; Yamaoka, Y.
Heterocycles 2021, 103, 177-182.
DOI: 10.3987/COM-20-S(K)6 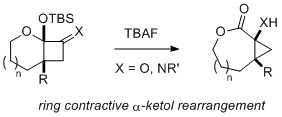
2020
- Synthetic Study on Acremoxanthone A, Part 2: Model Study on EFG Xanthone Moiety via Nitrile Oxide Cycloaddition–SNAr Sequence
Nakakohara, H.; Hirano, Y.; Ohmori, K.; Takikawa, H.; Suzuki, K.
Synlett 2020, 32, 423-428.
DOI: 10.1055/a-1303-5613
- Total Synthesis of (−)-Sigillin A: A Polychlorinated and Polyoxygenated Natural Product
Yamaoka, Y.; Nakayama, T.; Kawai, S.; Takasu, K.
Org. Lett. 2020, 22, 7721-7724.
DOI: 10.1021/acs.orglett.0c02930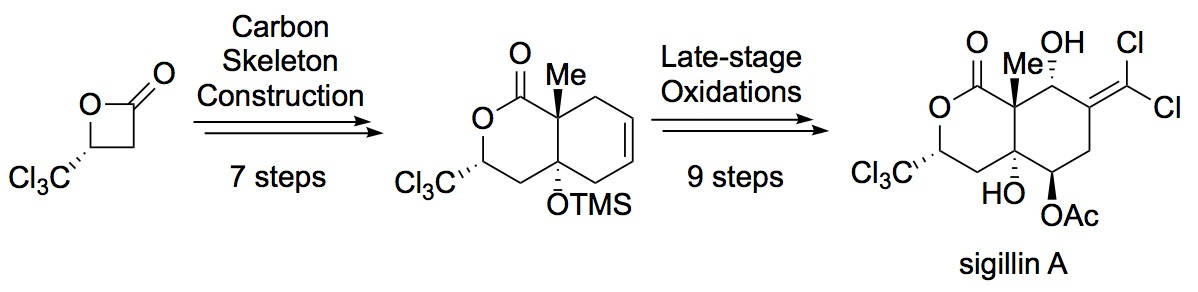
- CompRet: a comprehensive recommendation framework for chemical synthesis planning with algorithmic enumeration
Shibukawa, R.; Ishida, S.; Yoshizoe, K.; Wasa, K.; Takasu, K.; Okuno, Y.; Terayama, K.; Tsuda, K.
J. Cheminf. 2020, 12, 52.
DOI: 10.1186/s13321-020-00452-5
- Synthetic Study on Lactonamycins, Part 2: Stereoselective Access to ABCD-Ring System
Takikawa, H.; Murata, K.; Sato, S.; Kawada, T.; Nakakohara, H.; Ohmori, K.; Suzuki, K.
Synlett 2020, 31, 1623-1628.
DOI: 10.1055/s-0040-1707198
- Helical Nanographenes Embedded with Contiguous Azulene Units
Ogawa, N.; Yamaoka, Y.; Takikawa, H.; Yamada, K.; Takasu, K.
J. Am. Chem. Soc. 2020, 142, 13322-13327.
DOI: 10.1021/jacs.0c06156
京都大学プレスリリース
Chem-Stationスポットライトリサーチ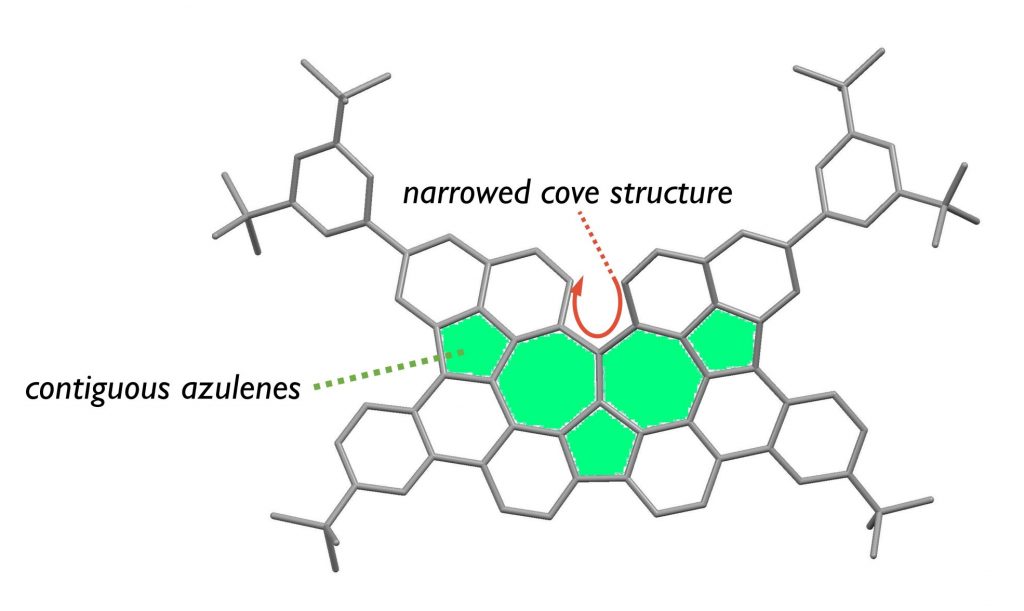
- Intramolecular Benzyne–Phenolate [4+2] Cycloadditions
Takikawa, H.; Nishii, A.; Takiguchi, H.; Yagishita, H.; Tanaka, M.; Hirano, K.; Uchiyama, M.; Ohmori, K.; Suzuki, K.
Angew. Chem. Int. Ed. 2020, 59, 12440-12444.
DOI: 10.1002/anie.202003131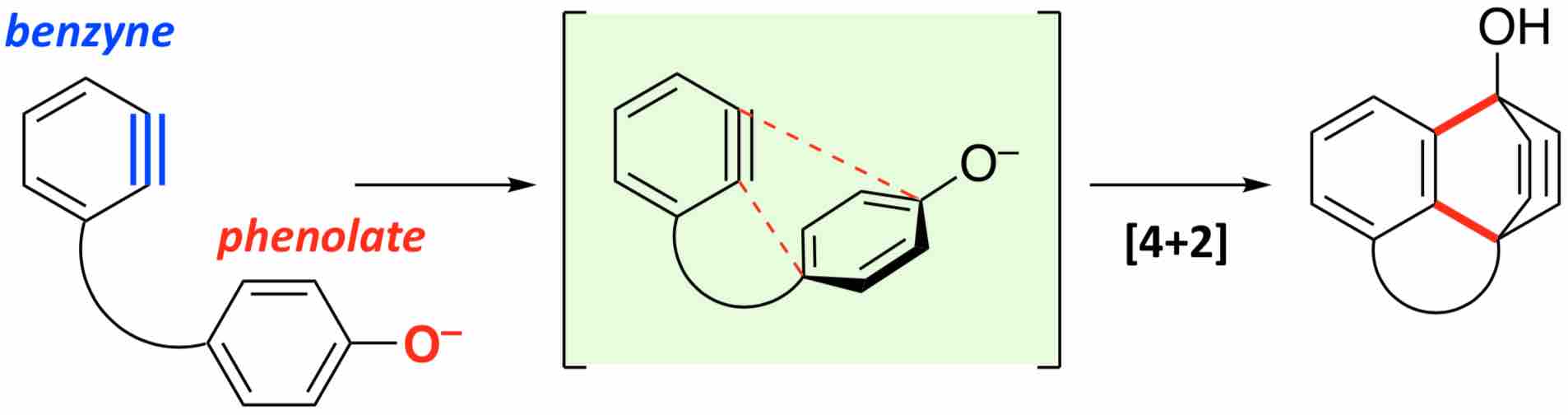
2019
- Prediction and Interpretable Visualization of Retrosynthetic Reactions using Graph Convolutional Networks
Ishida, S.; Terayama, K.; Kojima, R.; Takasu, K.; Okuno, Y.
J. Chem. Inf. Model. 2019, 59, 5026-5033.
DOI: 10.1021/acs.jcim.9b00538 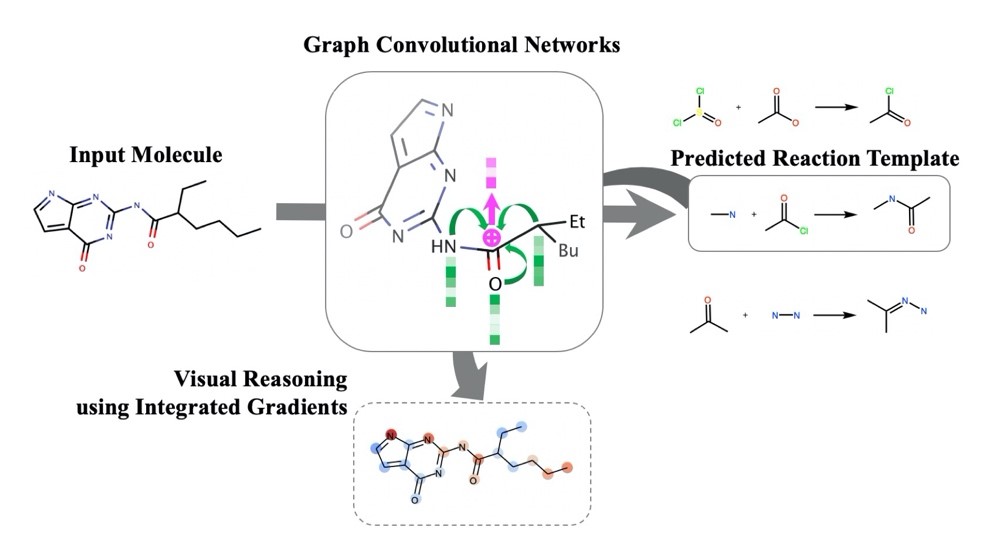
- Synthesis of Polycyclic Spirocarbocyles via Acid-Promoted Ring-Contraction/Dearomative Ring-Closure Cascade of Oxapropellanes
Ogawa, N.; Yamaoka, Y.; Takikawa, H.; Takasu, K.
Org. Lett. 2019, 21, 7563-7567.
DOI: 10.1021/acs.orglett.9b02835 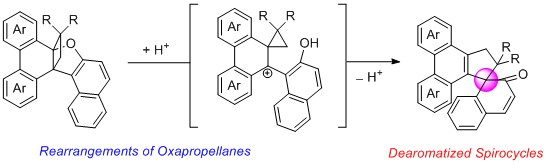
- Synthesis of Dibenzoxazonines by Domino (2+2) Cycloaddition—4π Electrocyclic Ring Opening Reaction of Cyclic Imines with Ynamides
Takasu, K.; Tsustumi, M.; Ito, T.; Takikawa, H.; Yamaoka, Y.
Heterocycles, 2020, 101, 423-428.
DOI: 10.3987/COM-19-S(F)39 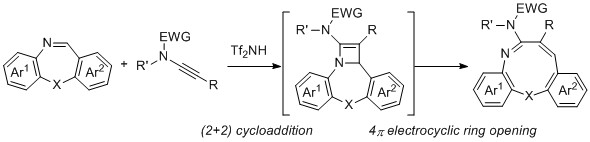
- Total Syntheses of Allelopathic 4-Oxyprotoilludanes, Melleolides and Echinocidins
Shimoda, K.; Yamaoka, Y.; Yoo, D.; Yamada, K.; Takikawa, H.; Takasu, K.
J. Org. Chem. 2019, 84, 11014-11024.
DOI: 10.1021/acs.joc.9b01589 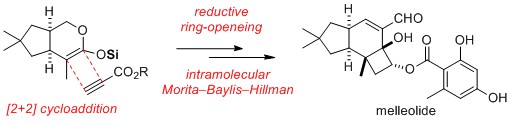
- Synthesis of Functionalized Medium-Sized trans-Cycloalkenes by 4π Electrocyclic Ring Opening– Alkylation Cascade
Ito, T.; Tsutsumi, M.; Yamada, K.; Takikawa, H.; Yamaoka, Y.; Takasu, K.
Angew. Chem. Int. Ed. 2019, 58, 11836-11840.
DOI: 10.1002/anie.201906665 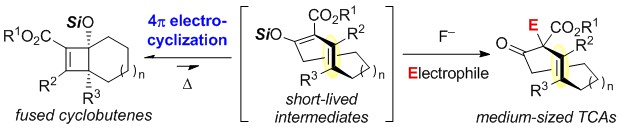
- Rapid Assembly of Protoilludane Skeleton through Tandem Catalysis: Total Synthesis of Paesslerin A and Its Structural Revision
Mogi, Y.; Inanaga, K.; Tokuyama, H.; Ihara, M.; Yamaoka, Y.; Yamada, K.; Takasu, K.
Org. Lett. 2019, 21, 3954–3958.
DOI: 10.1021/acs.orglett.9b01089 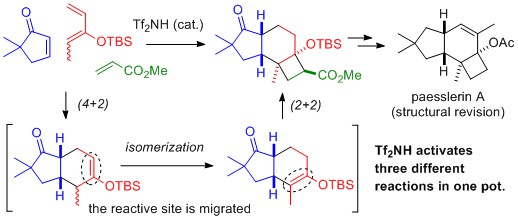
- Asymmetric Formal Synthesis of (+)-Catharanthine via Desymmetrization of Isoquinuclidine
Kono, M.; Harada, S.; Nozaki, T.; Hashimoto, Y.; Murata, S.; Gröger, H.; Kuroda, Y.; Yamada, K.; Takasu, K.; Hamada, Y.; Nemoto, T.
Org. Lett. 2019, 21, 3750–3754.
DOI: 10.1021/acs.orglett.9b01198 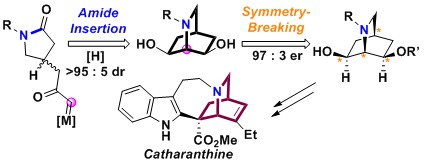
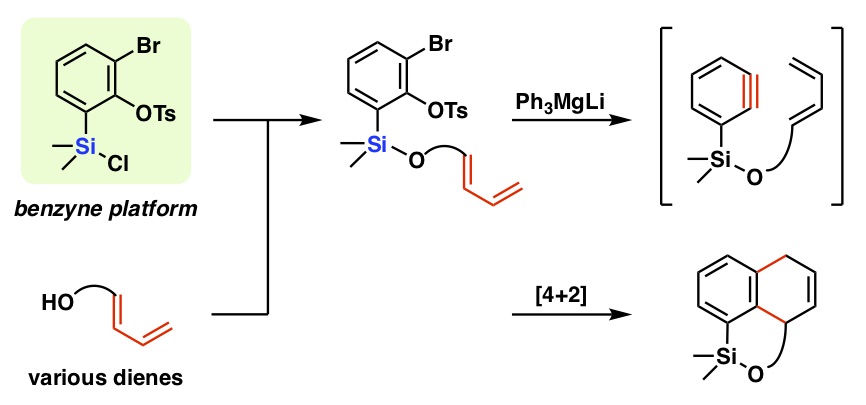
- 2-Bromo-6-(chlorodiisopropylsilyl)phenyl tosylate as an efficient platform for intramolecular benzyne–diene [4+2] cycloaddition
Nishii, A.; Takikawa, H.; Suzuki, K.
Chem. Sci. 2019, 10, 3840-3845.
DOI: 10.1039/c8sc05518a
- Optical Resolution via Catalytic Generation of Chiral Auxiliary
Kiyama, H.; Inokuma, T.; Kuroda, Y.; Yamaoka, Y.; Takasu, K.; Yamada, K.
Tetrahedron Lett. 2019, 60, 175-177.
DOI: 10.1016/j.tetlet.2018.12.006 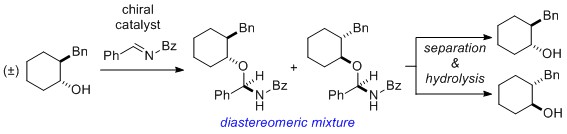
2018
- Silyl enol etherification by a Tf2NH/amine co-catalytic system for minimizing hazardous waste generation
Kurahashi, K.; Yamaoka, Y.; Takemoto, Y.; Takasu, K.
React. Chem. Eng. 2018, 3, 626-630.
DOI: 10.1039/C8RE00039E 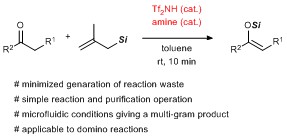
- Synthesis and Properties of Tribenzocarbazoles via an Acid-Promoted Retro (2+2)-Cycloaddition of Azapropellanes
Ogawa, N.; Yamaoka, Y.; Takikawa, H.; Tsubaki, K.; Takasu, K.
J. Org. Chem. 2018, 83, 7994-8002.
DOI: 10.1021/acs.joc.8b00870
Highlighted in Synfacts 
- Total Synthesis of Phenanthroquinolizidine Alkaloid Cryptopleurine and Phenanthroindolizidine Alkaloid Tylophorine
Yamaoka, Y.; Taniguchi, M.; Yamada, K.; Takasu, K.
Heterocycles 2018, 97, 292-305.
DOI: 10.3987/COM-18-S(T)19 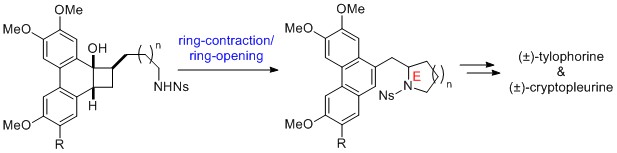
- Total Synthesis of Bis-anthraquinone Antibiotic BE-43472B
Yamashita, Y.; Hirano, Y.; Takada, A.; Takikawa, H.; Suzuki, K.
Synthesis 2018, 50, 2490-2515.
DOI: 10.1055/s-0037-1610136
2017
- Synthesis of Multi-substituted Cyclobutenes: Cyclic Strategy for (2+2) Cycloaddition of Ketene Silyl Acetals with Propiolates
Yamaoka, Y.; Ueda, M.; Yamashita, T.; Shimoda, K.; Yamada, K.; Takasu, K.
Tetrahedron Lett. 2017, 58, 2944-2947.
DOI: 10.1016/j.tetlet.2017.06.039 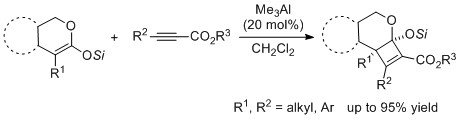
- Synthesis of π-Extended Fluoranthenes via a KHMDS-Promoted Anion and Radical Reaction Cascade
Ogawa, N.; Yamaoka, Y.; Yamada, K.; Takasu, K.
Org. Lett. 2017, 19, 3327-3330.
DOI: 10.1021/acs.orglett.7b01538 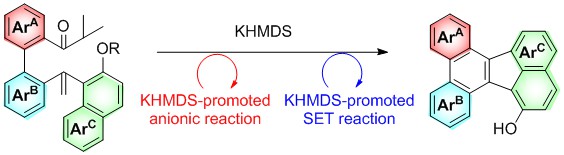
- Synthesis and biological evaluation of steroidal derivatives bearing a small ring as vitamin D receptor agonists
Arichi, N.; Fujiwara, S.; Ishizawa, M.; Makishima, M.; Hua, D. H.; Yamada, K.; Yamaoka, Y.; Takasu, K.
Bioorg. Med. Chem. Lett. 2017, 27, 3408-3411.
DOI: 10.1016/j.bmcl.2017.05.089 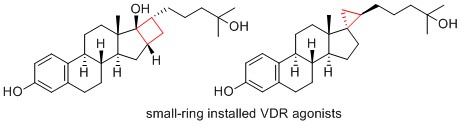
- Site-selective benzoin-type cyclization of unsymmetrical dialdoses catalyzed by N-heterocyclic carbenes for divergent cyclitol synthesis
Kang, B.; Wang, Y.; Kuwano, S.; Yamaoka, Y.; Takasu, K.; Yamada, K.
Chem. Commun. 2017, 53, 4469-4472.
DOI:10.1039/C7CC01191A 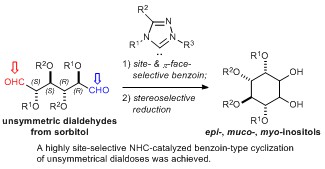
- Total Synthesis of (-)-Histrionicotoxin through a Stereoselective Radical Translocation-Cyclization Reaction
Sato, M.; Azuma, H.; Daigaku, A.; Sato, S.; Takasu, K.; Okano, K.; Tokuyama, H.
Angew. Chem. Int. Ed. 2017, 56, 1087-1091.
DOI: 10.1002/anie.201609941 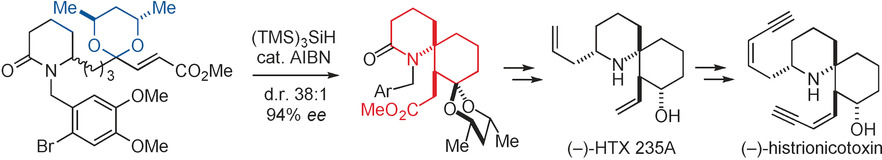
- Phosphine-Promoted Migrative Cyclization of Sulfonylalkynol and Sulfonylalkynamide for the Synthesis of Oxa- and Azacycles
Wang, Y.; Oriez, R.; Oh, S.; Miyakawa, Y.; Yamaoka, Y.; Takasu, K.; Yamada, K.
Heterocycels, 2017, 95, 314-321.
DOI: 10.3987/COM-16-S(S)22 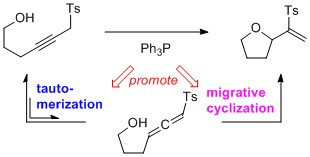
2016
- Use of a Catalytic Chiral Leaving Group for Asymmetric Substitutions at sp3-Hybridized Carbon Atoms: Kinetic Resolution of β-Amino Alcohols by p-Methoxybenzylation
Kuroda, Y.; Harada, S.; Oonishi, A.; Kiyama, H.; Yamaoka, Y.; Yamada, K.; Takasu, K.
Angew. Chem. Int. Ed. 2016, 55, 13137-13141.
DOI: 10.1002/anie.201607208 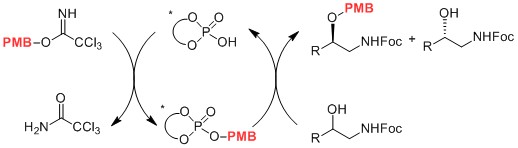
- Desymmetrization of acid anhydride with asymmetric esterification catalyzed by chiral phosphoric acid
Yamada, K.; Oonishi, A.; Kuroda, Y.; Harada, S.; Yamaoka, Y.; Kiyama, H.; Takasu, K.
Tetrahedron Lett. 2016, 57, 4098-4100.
DOI: 10.1016/j.tetlet.2016.07.093 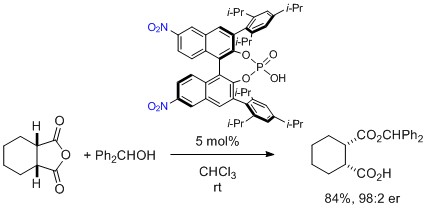
- Striking Difference between Succinimidomethyl and Phthalimidomethyl Radicals in Conjugate Addition to Alkylidenemalonate Initiated by Dimethylzinc
Yamada, K.; Matsumoto, Y.; Fujii, S.; Konishi, T.; Yamaoka, Y.; Takasu, K.
J. Org. Chem. 2016, 81, 3809-3817.
DOI: 10.1021/acs.joc.6b00485 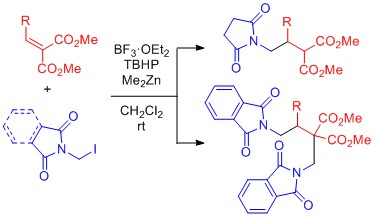
- Oxa- and Azacycle Formation via Migrative Cyclization of Sulfonylalkynol and Sulfonylalkynamide with N-Heterocyclic Carbene
Wang, Y.; Oriez, R.; Kuwano, S.; Yamaoka, Y.; Takasu, K.; Yamada, K.
J. Org. Chem. 2016, 81, 2652-2664.
DOI: 10.1021/acs.joc.6b00182 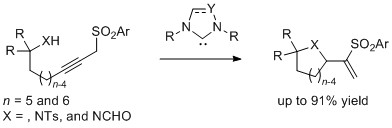
2015
- An Arylative Ring Expansion Cascade of Fused Cyclobutenes via Short-Lived Intermediates with Planar Chirality
Arichi, N.; Yamada, K.; Yamaoka, Y.; Takasu, K.
J. Am. Chem. Soc. 2015, 137, 9579-9682.
DOI: 10.1021/jacs.5b06576 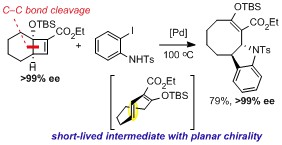
- Asymmetric Total Synthesis of Tylophorine via a Formal [2+2] Cycloaddition Followed by Migrative Ring Opening of a Cyclobutane
Yamaoka, Y.; Taniguchi, M.; Yamada, K.; Takasu, K.
Synthesis 2015, 47, 2819-2825.
DOI: 10.1055/s-0034-1380430
The 10 most popular articles of the last month. 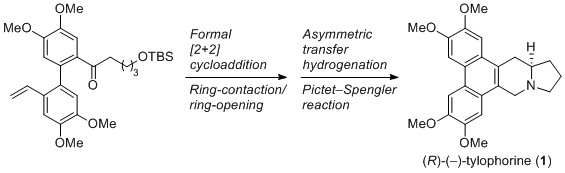
- Organocatalytic Activation of the Leaving Group in the Intramolecular Asymmetric SN2′ Reaction
Kuroda, Y.; Harada, S.; Oonishi, A.; Yamaoka, Y.; Yamada, K.; Takasu, K.
Angew. Chem. Int. Ed. 2015, 54, 8263-8266.
DOI: 10.1002/anie.201502831
Featured by Synfacts, selected as Synfact of the month, Featured by Advances in Engineering. 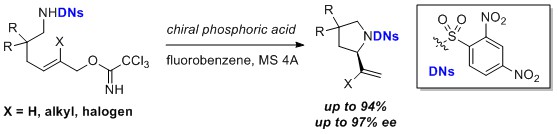
- The Development of a Brønsted Acid-promoted Ene-Ynamide Cyclization toward the Total Syntheses of Marinoquinolines A and C, and Aplidiopsamine
A, Yamaoka, Y.; Yoshida, T.; Shinozaki, M.; Yamada, K.; Takasu, K.
J. Org. Chem. 2015, 80, 957-964.
DOI: 10.1021/jo502467m
Featured by Synfacts 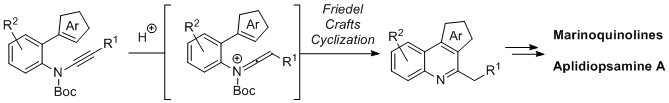
- Synthesis of Steroidal Derivatives Bearing a Small Ring Using a Catalytic [2+2] Cycloaddition and a Ring-Contraction Rearrangement
Arichi, N.; Hata, K.; Takemoto, Y.; Yamada, K.; Yamaoka, Y.; Takasu, K.
Tetrahedron 2015, 71, 233-243.
DOI: 10.1016/j.tet.2014.11.065 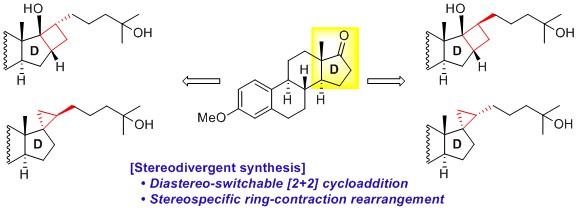
- Contiguous Radical Pivaloyloxymethylation-Directed C(sp3)-H Iodination of N-Tosyl Cycloalkanecarbaldimine
Fujii, S.; Nakano, M.; Yamaoka, Y.; Takasu, K.; Yamada, K.; Tomioka, K.
Tetrahedron Lett. 2015, 56, 3086-3089.
DOI: 10.1016/j.tetlet.2014.11.105 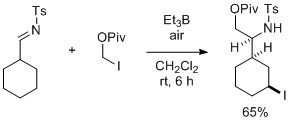
- Hydrostannylation-Cross-Coupling Strategy for Stereoselective Synthesis of Alkylidenemalonates and Related α,β-Unsaturated Esters
Fujiwara, S.; Cadou, R.; Yamaoka, Y.; Takasu, K.; Yamada, K.
Eur. J. Org. Chem. 2015, 1264-1272.
DOI: 10.1002/ejoc.201403429 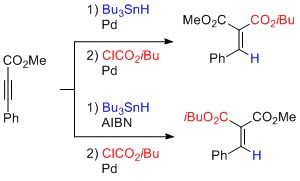
- Synthetic Studies toward Penitrem E: Enantiocontrolled Construction of B-E Rings
Yoshii, Y.; Otsu, T.; Hosokawa, T.; Takasu, K.; Okano, K.; Tokuyama, K.
Chem. Commun. 2015, 51, 1070-1073.
DOI:10.1039/C4CC08505A
Featured by Synfacts 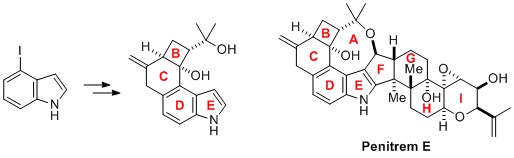
- N-Heterocyclic Carbene-Catalyzed Benzoin Strategy for Divergent Synthesis of Cyclitol Derivatives from Alditols
Kang, B.; Suto, T.; Wang, Y.; Kuwano, S.; Yamaoka, Y.; Takasu, K.; Yamada, K.
Adv. Synth. Catal. 2015, 357, 131-147.
DOI: 10.1002/adsc.201400712 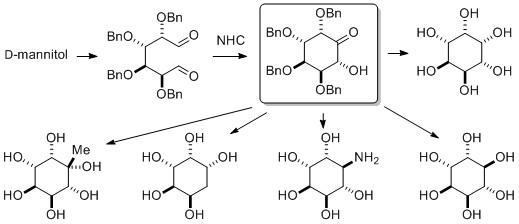
2014
- Stereocontrolled Total Synthesis And Biological Evaluation of (-)- And (+)-Petrosin and Its Derivatives
Toya, H.; Satoh, T.; Okano, K.; Takasu, K.; Ihara, M.; Takahashi, A.; Tanaka, H.; Tokuyama, H.
Tetrahedron 2014, 70, 8129-8141.
DOI: 10.1016/j.tet.2014.08.009 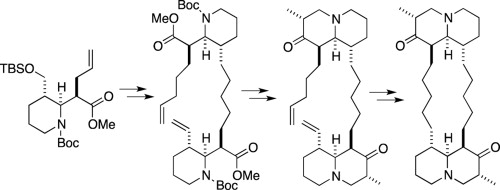
- Radical Amidomethylation of Imines
Fujii, S.; Konishi, T.; Matsumoto, Y.; Yamaoka, Y.; Takasu, K.; Yamada, K.
J. Org. Chem. 2014, 79, 8128-8133.
DOI: 10.1021/jo501332j 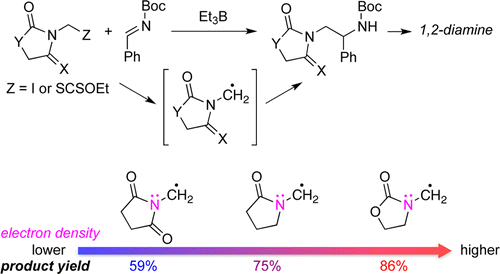
- Equilibration of the EtAlCl2-Catalyzed [2 + 2] Cycloaddition of Silyl Enol Ethers: Diastereoselectivity Switch in the Synthesis of Fused Cyclobutanes
Hata, K.; Arichi, N.; Yamaoka, Y.; Yamada, K.; Takemoto, Y.; Takasu, K.
Asian J. Org. Chem. 2014, 3, 706-710.
DOI: 10.1002/ajoc.201402042 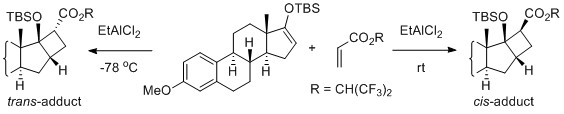
- Synthesis of Functionalized Polycyclic Aromatic Compounds via a Formal [2+2]-cycloaddition
Nagamoto, Y.; Yamaoka, Y.; Fujimura, S.; Takemoto, Y.; Takasu, K.
Org. Lett. 2014, 16, 1008-1011.
DOI: 10.1021/ol403757e 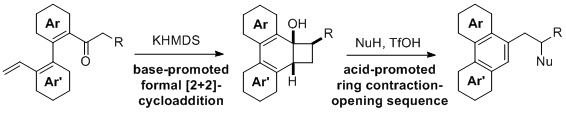
2013
- Enhanced Rate and Selectivity by Carboxylate Salt as a Basic Co-catalyst in Chiral N-Heterocyclic Carbene-Catalyzed Asymmetric Acylation of Secondary Alcohols
Kuwano, S.; Harada, S.; Kang, B.; Raphael, O.; Yamaoka, Y.; Takasu, K.; Yamada, K.
J. Am. Chem. Soc. 2013, 135, 11485-11488.
DOI: 10.1021/ja4055838
Featured by Synfacts 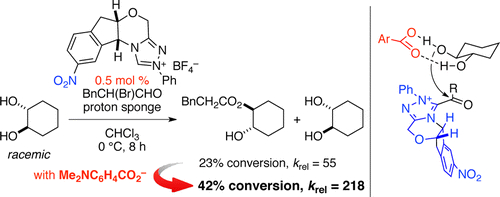
- Kinetic resolution of secondary alcohols catalyzed by chiral phosphoric acids
Harada, S.; Kuwano, S.; Yamaoka, Y.; Yamada, K.; Takasu, K.
Angew. Chem. Int. Ed. 2013, 52, 10227-10230.
DOI: 10.1002/anie.201304281
Featured by Synfacts 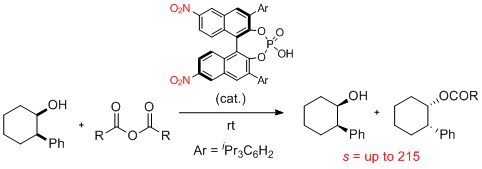
- Synthesis of 2,3,4,5-Tetra-substituted Pyrroles via a Base-Promoted Double Michael Reaction of Oxime-enoates with Nitroolefins
Kuroda, Y.; Imaizumi, K.; Yamada, K.; Yamaoka, Y.; Takasu, K.
Tetrahedron Lett. 2013, 54, 4073-4075.
DOI: 10.1016/j.tetlet.2013.05.100 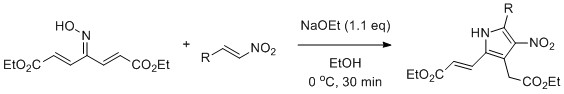
- Selective Synthesis of Poly-Substituted Dihydroquinolines and α,β-Unsaturated Amidines by A Catalytic Reaction of Ynamides with Ketimines
Kuroda, Y.; Shindoh, N.; Takemoto, Y.; Takasu, K.
Synthesis 2013, 45, 2318-2326.
DOI: 10.1055/s-0033-1339191 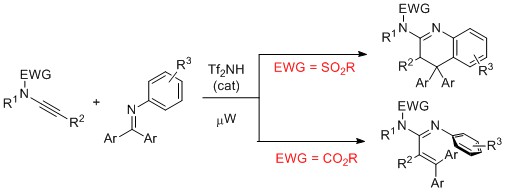
- Critical profiles of chiral diether-mediated asymmetric conjugate aminolithiation of enoate with lithium amide as a key to the total synthesis of (-)-kopsinine
Harada, S.; Sakai, T.; Takasu, K.; Yamada, K.; Tomioka, K.
Tetrahedron 2013, 69, 3264-3273.
DOI: 10.1016/j.tet.2013.02.035 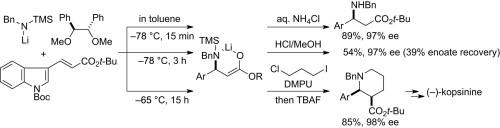
- pH-sensitive DNA cleaving agents: in situ activation by ring contraction of benzo-fused cyclobutanols
Nagamoto, Y.; Hattori, A.; Kakeya, H.; Takemoto, Y.; Takasu, K.
Chem. Commun. 2013, 49, 2622-2624.
DOI: 10.1039/c3cc39246e 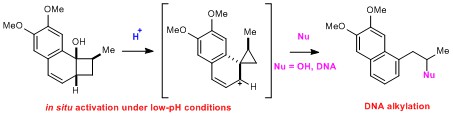
- Study of Ring-Opening Reaction of Spiro[5.2]octenes with Aqueous Hydro-halic Acid: Substituent Effect on the Regioselectivity
Nagamoto, Y.; Takemoto, Y.; Takasu, K.
Synlett 2013, 24, 120-124.
DOI: 10.1055/s-0032-1317745 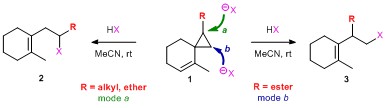
2012
- Total Synthesis of (+)-trans-Dihydronarciclasine Utilizing Asymmetric Conjugate Addition
Yamada, K.; Mogi, Y.; Mohamed, M. A.; Takasu, K.; Tomioka, K.
Org. Lett. 2012, 14, 5868-5871.
DOI: 10.1021/ol302757y 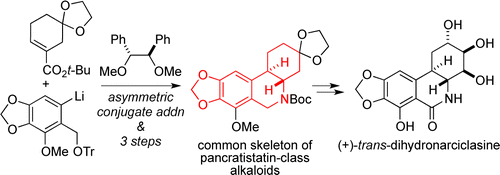
- Total Synthesis of (-)-Kopsinine by an Asymmetric One-Pot [N+2+3] Cyclization
Harada, S.; Sakai, T.; Takasu, K.; Yamada, K.; Tomioka, K.
Chem. Asian J. 2012, 7, 2196-2198.
DOI: 10.1002/asia.201200575 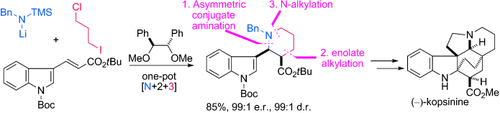
- General Entry to Asymmetric One-Pot [N + 2 + n] Cyclization for the Synthesis of Three- to Seven-Membered Azacycloalkanes
Harada, S.; Sakai, T.; Takasu, K.; Yamada, K.; Tomioka, K.
J. Org. Chem. 2012, 77, 7212-7222. Featured by Synfacts
DOI: 10.1021/jo301495a 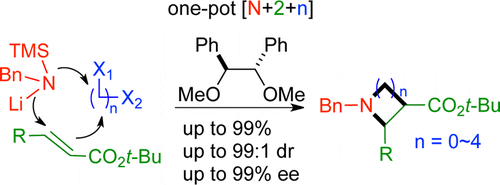
- Facile Isomerization of Silyl Enol Ethers Catalyzed by Triflic Imide and Its Application to One-pot Isomerization-(2 + 2) Cycloaddition
Inanaga, K.; Ogawa, Y.; Nagamoto, Y.; Daigaku, A.; Tokuyama, H.; Takemoto, Y.; Takasu, K.
Beilstein J. Org. Chem. 2012, 8, 658-661.
DOI: 10.3762/bjoc.8.73 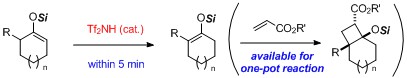
- Room-temperature, acid-catalyzed [2+2] cycloadditions: Suppression of side reactions by using a flow microreactor system
Kurahashi, K.; Takemoto, Y.; Takasu, K.
ChemSusChem 2012, 5, 270-273.
DOI: 10.1002/cssc.201100373 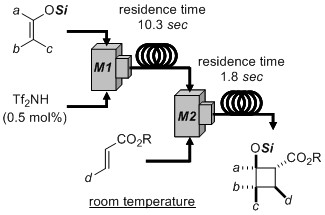
2011
- Formal (3 + 3) Cycloaddition of Silyl Enol Ethers Catalyzed by Trifric Imide: Domino Michael Addition-Claisen Condensation Accompanied with Isomerization of Silyl Enol Ethers
Azuma, T.; Takemoto, Y.; Takasu, K.
Chem. Pharm. Bull. 2011, 59, 1190-1193.
DOI: 10.1248/cpb.59.1190 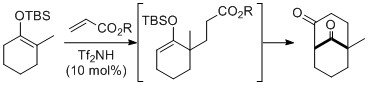
- Catalyst-Controlled Torquoselectivity Switch in the 4π Ring-Opening Reaction of 2-Amino-2-azetines Giving β-Substituted α,β-Unsaturated Amidines
Shindoh, N.; Kitaura, K.; Takemoto, Y.; Takasu, K.
J. Am. Chem. Soc. 2011, 133, 8470-8473.
DOI: 10.1021/ja202576e 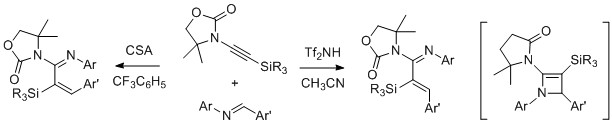
- Unprecedented Synthesis of N,N-Divinylamines by Tf2NH-Catalyzed Reaction of Ynamide with Ketimine
Shindoh, N.; Takemoto, Y.; Takasu, K.
Heterocycles 2011, 82, 1133-1136.
DOI: 10.3987/COM-10-S(E)87 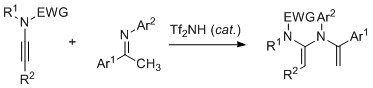
- Gold(I)-Catalyzed Polycyclizations of Polyenyne-Type Anilines Based on Hydroamination and Consecutive Hydroarylation Cascade
Hirano, K.; Inaba, Y.; Takasu, K.; Oishi, S.; Takemoto, Y.; Fujii, N.; Ohno, H.
J. Org. Chem. 2011, 76, 9068-9080.
DOI: 10.1021/jo2018119
Reviews
- 脱芳香化された多環性芳香族ポリケチド天然物の合成戦略
瀧川紘,鈴木啓介,
有機合成化学協会誌,2019, 77(1), 13-25.
DOI: 10.5059/yukigoseikyokaishi.77.13
- Aryne-Based Strategy in Total Synthesis of Naturally-Occurring Polycyclic Compounds
Takikawa, H.; Nishii, A.; Sakai, T.; Suzuki, K.
Chem. Soc. Rev. 2018, 47, 8030-8056.
DOI: 10.1039/c8cs00350e
Top 5% of highly cited authors with RSC journals in 2019
- ポリケチド系多環性化合物の全合成における試行錯誤
瀧川紘,
有機合成化学協会誌,2018, 76(5), 478-481.
DOI: 10.5059/yukigoseikyokaishi.76.478
- キラルリン酸による触媒的不斉置換反応およびアシル化反応への展開
黒田悠介,原田慎吾,山田健一,高須清誠,
有機合成化学協会誌,2018, 76(4), 325-335.
DOI: 10.5059/yukigoseikyokaishi.76.325 
- Synthesis of Azaheterocycles and Related Molecules by Tf2NH-Catalyzed Cycloadditions
Shindo, N.; Takasu, K.
Heterocycles 2018, 96, 195-218.
DOI: 10.3987/REV-17-875 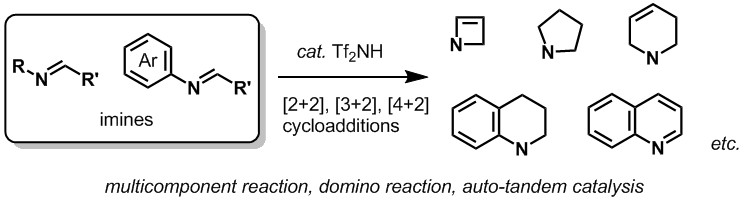
- 形式的[2+2]環化反応を用いた多環芳香族炭化水素の合成とその応用 (Efficient Synthesis of Polyaromatic Hydrocarbon via a Formal [2+2] Cycloaddition)
山岡庸介(Yousuke Yamaoka)
Yakugaku Zasshi, 2016, 136, 1517-1523.
DOI: 10.1248/yakushi.16-00202
- π-Delocalized Lipophilic Quaternary Cations as new Candidates for Antimalarial, Antitrypanosomal and Antileishmanial Agents: Synthesis, Evaluation of Antiprotozoal Potency and Insight of their Action Mechanism
Takasu, K.
Chem. Pharm. Bull. 2016, 64(7), 656-667.
DOI: 10.1248/cpb.c16-00234 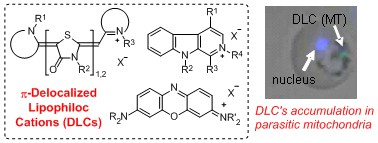
- Synthesis of Multisubstituted Silyloxy-based Donor-Acceptor Cyclobutanes by an Acid-Catalyzed [2+2] Cycloaddition
Takasu, K.
Israel J. Chem. 2016, 56(6-7), 488-498.
DOI: 10.1002/ijch.201500096 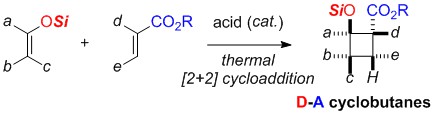
- トリフリックイミドのオート・タンデム触媒作用を利用する有機合成
高須清誠,
有機合成化学協会誌,2014, 72(7), 770-780.
DOI: 10.5059/yukigoseikyokaishi.72.770 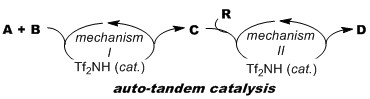
- 軸性不斉をもつアミジンの触媒的不斉合成法の開発とキラル合成素子としての活用
高須清誠,
薬学研究の進歩, 2011, 31-36.
Books
- フローマイクロ合成の最新動向
高須清誠著、ファインケミカル合成プロセス
シーエムシー出版, 2021, pp.102-110. ISBN 978-4-7813-1615-4
- スタンダード薬学シリーズII-8 薬学研究, 日本薬学会編
藤多哲朗、高須清誠著、人とのつながりが結実して誕生した新薬-冬虫夏草を起源とする多発性硬化症治療薬フィンゴリモド
東京化学同人, 2017, pp.36-44. ISBN 9784807917228
- くすりをつくる研究者の仕事 京都大学大学院薬学研究科編
高須清誠、第2章 薬を合成する-炭素の錬金術師
化学同人、2017, pp.35-62. ISBN 9784759819311.
- スタンダード薬学シリーズII -3 化学系薬学 II.
伊藤喬、石崎幸、石塚忠男、橘高敦、高須清誠 編
東京化学同人, 2016.
- スタンダード薬学シリーズII -3 化学系薬学 III.
伊藤喬、石崎幸、石塚忠男、橘高敦、高須清誠、供田洋、森田博史 編、
東京化学同人, 2016.
- スタンダード薬学シリーズII- 3 化学系薬学 I. 化学物質の性質と反応
伊藤喬、石崎幸、石塚忠男、橘高敦、高須清誠 編、第1章 化学結合と分子、
東京化学同人, 2015, pp.2-14.
- ウォーレン有機合成-逆合成からのアプローチ-(翻訳)
Warren, S. and Wyatt, P. 著、柴崎正勝、橋本俊一 監訳、金井求、木越英夫、高須清誠、松永茂樹 訳、
東京化学同人,2014, pp. 202-279.
- Catalytic [2+2] Cycloaddition of Silyl Enol Ethers
Yamaoka, Y. and Takasu, K.
in Methods and Applications of Cycloaddition Reactions in Organic Syntheses, ed. by Nishiwaki, N. Wiley, New York, 2014, pp.115-134.
DOI:10.1002/9781118778173.ch04
- Reactions involving an α,β-Unsaturated Carbonyl Compound as Electrophilic Component with [2+2] Cycloaddition as the Key Step
Takasu, K.
in Science of Synthesis (Multicomponent Reactions), 2013, Thieme, pp.195-210.
- Heterogeneous Reactions
Takasu, K.
in Microreactors in Organic Chemistry and Catalysis, 2nd edition, ed by Wirth, T. Wiley-VCH, Weinheim, 2013, pp. 151-196.
- 原虫オルガネラの固有環境認識を基盤とした抗マラリア活性物質の創製
高須清誠,
ナノバイオ技術と創薬研究(佐治英郎ら編),メディカルドゥ,2012、pp.72-77.
- Thiourea, N-[3,5-Bis(trifluoromethyl)phenyl]-N’-[(1R,2R)-2-(dimethylamino)cyclohexyl]-
Takasu, K.; Takemoto, Y.
Encyclopedia of Reagents for Organic Synthesis (Wiley), 2012, 1-4.
DOI: 10.1002/047084289X.rn01414 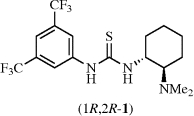
Patents
- 高須清誠、奥野恭史、瀬木恵里、原貴史、藤村駿、山本佳宏、金井千里、吉川達也、閨正博、河野晋哉 「ピペラジン誘導体およびその用途」 WO 2014/024993(特願JP2012-177572)
- 高須清誠,竹本佳司,倉橋慧 「シリルエノール類の製造法」 特願2010-48290
- 高須清誠,井原正隆,稲永風人 「多置換シクロブタン及び多置換シクロブテン化合物の製造方法」 特開2007-119488
- 井原正隆,高須清誠,Pudhom Khanitha,北口博司,川上雅之,佐藤幸藏 「アザロダシアニン化合物を有効成分として含有する原虫感染症治療剤」 WO2006/137258 (特願JP2005-50064)
- 井原正隆,高須清誠,北口博司,川上雅之,佐藤幸藏 「フェノキサジニウム化合物を有効成分とする原虫感染症治療剤」 WO2006/087935 (特願JP2005-41238)
- 高須清誠,井原正隆,稲永風人 「多置換シクロブタン及び多置換シクロブテン化合物の製造方法」 WO2006/009119 (特願JP2006-529195)
- 井原正隆,高須清誠,Pudhom Khanitha,北口博司,川上雅之,佐藤幸藏 「原虫寄生感染症の予防又は治療用医薬組成物」 特開2006-104116
- 井原正隆,高須清誠,Pudhom Khanitha,北口博司,川上雅之,佐藤幸藏 「抗トリパノソーマ剤」 特開2006-104115,未請求
- 井原正隆,高須清誠 「マラリア増殖阻害に関与するタンパク質」 特開2006-67867
- 井原正隆,高須清誠,西田直子 「置換ピペリジン及びその製法」 特開JP2005-75785
- 井原正隆,高須清誠,寺内広毅,関田節子,高橋真理衣 「リーシュマニア治療剤」 特開JP2004-331545
- 井原正隆,高須清誠,寺内広毅 「メロシアニン系色素化合物及びロダシアニン系色素化合物のコンビナトリアル製造方法」 特開JP2004-137271
- 井原正隆,高須清誠,伊上博史,綿矢有佑,金惠淑 「マラリア感染診断剤およびマラリア原虫染色剤」 特開JP2004-144526
- 井原正隆,高須清誠,綿矢有佑,金惠淑 「ロダシアニン系色素化合物を含有する抗マラリア剤」 特開JP2003-34642
- 井原正隆,高須清誠,綿矢有佑,金惠淑 「四環性複素化合物を含有する抗マラリア剤」 特開JP2003-34641
- 井原正隆,高須清誠,綿矢有佑,金惠淑 「ロダシアニン色素化合物を含有する抗マラリア剤」 特開JP2003-34640
- 井原正隆,高須清誠,綿矢有佑,金惠淑 「抗マラリア活性を有する新規化合物」 特開JP2001-24755
































































































